There’s nothing new under the sun. And no free lunch. You can’t get something for nothing or squeeze blood from a rock. But sometimes, very occasionally, you get what you need. After we decided that we weren’t going to hook up to the natural gas pipeline, effectively making this an all-electric house, what we needed is the best possible way to use electricity.
In the last five years a new class of heating devices has been developed. High efficiency heat pumps use a technology that has been around for a long time: as long as refrigerators and air conditioners. The latest generation of heat pumps is more efficient and can make heat in more extreme circumstances (read: cold). Now heat pumps can heat a home all season long, and the cost to run a heat pump is close to the cost to heat with home heating oil: much less than heating with propane; a little more than heating with natural gas. I’m on my way to explaining about our heat pump water heater, which puts the heat into water instead of into the air, but before I get there, let’s take a lap around Physics.
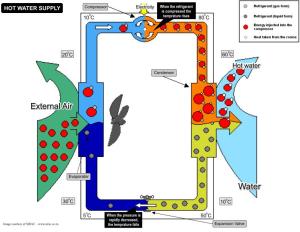
By manipulating the pressure of refrigerant, heat pumps move energy from one place to another. Sometimes the energy is low-grade, like cold air. But the heat pump turns it into high-grade energy by moving and concentrating it.
Heat pumps take advantage of a funny loophole in physics: phase change. Phase change happens when any substance goes to a higher or lower energy level, for instance:
- water turning to ice
- perfume evaporating into the air
- steel melting
Between the three phases, from lowest- to highest-energy level: solid-liquid-gas, a tremendous amount of energy is moved around. With something like steel it’s easy to picture the energy required to make the steel go from a solid to a liquid. The heat that goes into solid steel is energy that is used to drive the phase change to liquid. Oddly, a substance can remain the same temperature while either absorbing or releasing large amounts of energy if the substance is going through a phase change. Consider water. You can have 32 deg F water and you can have 32 deg F ice. When ice melts it absorbs a lot of energy from its environment, but it doesn’t warm up; it just changes phase. In the other direction, liquid water can turn to ice, giving up a lot of energy in order to do so, but without getting any colder.
Engineers, maybe it was Trane, the inventor of the modern air conditioner, figured out a way to manipulate phase change by forcing a substance to condense at a temperature when it would normally be a gas. That’s kind of like turning 50 degree water into ice. Some substances are more easily manipulated in this way, and will phase change at temperatures comfortable to humans. These are the refrigerants. There are a lot of different refrigerants and some of them are really bad for the environment, notably thinning out the ozone layer to the point that it’s pretty well swiss-cheesed. (On a side note, one of Nik’s clients told us last night that by pure, dumb luck humans avoided a very rapid extinction when Dupont decided to use chlorine in refrigerants instead of bromine. They act and cost the same, but Bromine would have utterly destroyed the ozone in a couple of years, making the Earth uninhabitable for most plants and animals.)
Here’s the refrigerant cycle in a nutshell, and in 5th grade vocabulary. I always think of physics in terms that I grasped when I was in 5th grade. The refrigerant, which is normally gas at room temperature and above, starts as a gas. It gets pumped into a compressor, which is the part of the whole system that uses big electricity, where it gets squeezed until it turns into a liquid. Substances phase change at a given temperature and pressure. If you change the pressure you can get the stuff to phase change at an odd temperature. That’s the whole trick about heat pumps. The liquid is pumped to an evaporator where it’s allowed to turn back into a gas, thereby absorbing a lot of heat (usually out of the air, but sometimes water). When the refrigerant evaporates into a gas it cools air (or water) around it. The warm, gaseous refrigerant is pumped back to the compressor where it goes through all that squeezing again. It’s a cycle.
The way I’ve described it, the system is doing cooling. However, the whole process can be reversed and then the system becomes a heater. That’s what a heat pump is: a reversible air conditioner. When you reverse the flow of the refrigerant through the compressor the refrigerant goes into the house as a gas. Gaseous refrigerant condenses, giving up heat to the air (or water). Viola! a heater instead of an air conditioner. In cooling mode, the condensing happens outside, “ejecting” heat to the back yard; in heating mode the heat is “ejected” inside the house. In heating mode, energy (or heat if you prefer to think of it that way) is absorbed from outdoors and moved into the house; in cooling mode energy is absorbed from within the house and moved to the back yard. Refrigerants and compressor are just a way of moving energy around.
And that is how heat pumps got their reputation as being “free heat”. The energy that heats your house is indeed energy that came from outdoor air or ground water. Even at low temperatures air holds enough energy to heat a home. Of course, you need a lot of air to do that, and the heat pump uses a lot of electricity to make it happen. It’s kind of like getting something for free but you have to pay the shipping. Heat pumps also have a reputation as being “renewable” because the heat is energy that is supplied by the planet, but again you have to use a lot of electricity to get that heat, and since most electricity is not renewably generated it’s misleading to say so. Nonetheless, it is a neat trick, and with recent improvements to heat pumps it works even in Vermont!

Great explanation! Maybe when you want a new project you can start a whole science series called “Science in 5th grade language.” I’d read it.
By: Noah&Amanda on May 10, 2013
at 5:16 pm
[…] where our wetness comes from. Our dehumidifier removes moisture from the air by the same refrigerant cycle I described for the heat pump. The dehumidifier we brought with us from our other house pooped […]
By: We Need a New Dehumidifier | Energy Freak Show on July 7, 2013
at 7:15 pm
[…] dust on the fins in the heat pump that makes our hot water. For a primer on heat pumps, see this post. The fins are where energy in the air is transferred to the refrigerant. A lot of air has to […]
By: Dust Blows | Energy Freak Show on February 5, 2017
at 7:36 pm